February 2019, Volume 28, Issue 2, pp 434–441| Cite as
Krappinger, D., Lindtner, R.A., Zegg, M.J. et al. Eur Spine J (2019) 28: 434.
https://doi.org/10.1007/s00586-018-5796-5
To revisit the concept of spondylotic traumatic central cord syndrome (TCCS) by assessing the frequency of discoligamentous injury and to correlate magnetic resonance imaging (MRI) and intraoperative findings.
A retrospective analysis of twenty-three consecutive patients (mean age: 62.7 ± 14.8 years) with profound spondylotic TCCS after acute cervical hyperextension trauma but without signs of instability on initial CT scans who underwent anterior surgical decompression and fusion was performed. Sensitivity and specificity of MRI in the detection of anterior longitudinal ligament disruption were calculated. The topographic relations between surgically verified segmental instabilities and spinal cord signals on MRI were analyzed. The cervical MRI scans of all patients were evaluated by the radiologist on call at time of admission, re-assessed by a specialized MRI radiologist for the purpose of this study and compared with intraoperative findings.
Intraoperative findings revealed 25 cervical spine segments with hyperextension instability in 22 of 23 (95.7%) patients. The radiologist on call correctly assessed segmental hyperextension instability in 15 of 25 segments (sensitivity: 0.60, specificity: 1.00), while the specialized MRI radiologist was correct in 22 segments (sensitivity: 0.88, specificity: 1.00). In 17 of 23 (73.9%) patients, the level of spinal cord signal on MRI matched the level of surgically verified segmental instability.
Our findings challenge the traditional concept of spondylotic TCCS as an incomplete cervical spinal cord injury without discoligamentous injury and emphasize the importance of MRI as well as the radiologist’s level of experience for the assessment of segmental instability in these patients.
These slides can be retrieved under Electronic Supplementary Material.

Central cord syndrome Hyperextension injury Discoligamentous injury Cervical spine injury Magnetic resonance imaging
The online version of this article ( https://doi.org/10.1007/s00586-018-5796-5) contains supplementary material, which is available to authorized users.
Traumatic central cord syndrome (TCCS) is the most frequent type of incomplete cervical spinal cord injury [1, 2, 3]. Typical symptoms of cervical TCCS are more severe motor impairment of the upper extremities compared to the lower extremities and varying degrees of sensory loss [4]. One pathognomonic symptom of TCCS is massive dysesthesia of the distal upper extremities, commonly described as “burning hands” by the patients [4, 5, 6, 7].
TCCS mainly occurs in three situations: (a) fractures or dislocations of the cervical spine, (b) acute traumatic cervical disc herniations, and (c) hyperextension injuries in patients with cervical stenosis and spondylosis without segmental instability (spondylotic TCCS) [4, 7, 8, 9]. In the first two situations, urgent surgical intervention with reduction, decompression, and stabilization is widely considered mandatory in order to promote neurological recovery [2, 10, 11, 12, 13, 14]. The role and timing of surgical interventions for spondylotic TCSS, however, remains unclear [3, 7]. There is no need for reduction and stabilization in the presence of a degenerative but stable cervical spine. Additionally, these patients frequently experience rapid neurologic recovery within the first hours after trauma [3, 12]. There is a lack of prospective or even randomized studies concerning indications for surgical intervention in spondylotic TCCS. An expert consensus paper recommends early decompression in patients with profound neurological impairment (American Spinal Injury Association (ASIA) Impairment Scale (AIS) grade C or worse) and initial observation in patients with less severe neurological impairment with optional surgery at a later date [7]. These recommendations, however, are based on the assumption of a stable spondylotic cervical spine without acute discoligamentous injury [3].
Spinal cord injury without evidence of fractures or discoligamentous injuries of the spine is known as SCIWORA (“Spinal Cord Injury Without Radiological Abnormalities”). SCIWORA is considered to mainly occur in pediatric patients [15, 16, 17]. The elasticity of the pediatric spine may allow to stretch to some degree before structural damage occurs, whereas the spinal cord is less flexible and elastic and therefore sustains traction damage leading to neurological impairment [18, 19]. In patients with spondylosis and without acute instability, however, the elasticity of the spinal column and the segmental range of motion are typically greatly reduced [8, 20]. It is debatable if the small residual range of motion without further instability is adequate to result in profound TCCS. The aim of the present study therefore was to revisit the concept of spondylotic TCCS using magnetic resonance imaging (MRI) and correlative intraoperative findings. Specifically, we sought to determine the rate of hyperextension instability in patients with profound TCCS (i.e., AIS grade C or worse) and to correlate MRI and intraoperative findings.
A consecutive series of 23 patients treated between 2011 and 2016 at a level 1 trauma center were included in this study. Inclusion criteria were: (1) history of an acute trauma of the cervical spine; (2) TCCS at admission; (3) availability of both computed tomography (CT) and MRI scans obtained at admission; (4) no evidence of fractures or signs of instability on the initial CT scan; (5) hyperintense spinal cord signal on T2-weighted and short tau inversion recovery (STIR) MRI scans; and (6) spinal cord decompression via ACDF within one week after trauma. Exclusion criteria comprised the following: (1) no history of an acute trauma of the cervical spine; (2) no neurological deficits or neurological deficits other than TCSS; (3) no CT and/or MRI scan at admission; (4) evidence of fractures or signs of instability on the initial CT scan; (5) no hyperintense spinal cord signal in initial T2-weighted and STIR MRI scans; and (6) nonoperative treatment or surgical treatment other than ACDF.
CT and MRI scans were performed immediately after admission. MRI scans were obtained using a 1.5 T system (Avanto Fit, Siemens Healthineers, Erlangen, Germany). T1- and T2-weighted images were acquired in sagittal and axial orientation, and an additional STIR sequence was performed in the sagittal plane. Fractures or signs of instability on the CT scan (i.e., subluxation and dislocation of the facet joints) were ruled out by the radiologist and the trauma surgeon on call. The radiologist on call further assessed the MRI scan and was aware of the clinical situation of the patient, i.e., the history of an acute trauma of the cervical spine and the neurological impairment. The radiologist’s report describing the cord signal as well as the status of the anterior (ALL) and posterior longitudinal ligament (PLL) were retrospectively analyzed. If the ALL was described as discontinuous by the radiologist on call, the anterior tension band was defined as ruptured, and the corresponding segment was defined radiologically as “unstable in extension” [21].
Surgery was performed within the first week after trauma in all patients by spine surgeons experienced in the treatment of cervical spine injuries. Indications for surgery followed institutional guidelines which were based on an expert consensus paper published in 2010 [7]. Surgery was performed in patients with an American Spinal Injury Association (ASIA) Impairment Scale (AIS) grade C or worse (as assessed on the day after trauma) within the next 24 h. In patients with AIS grade D and incomplete remission or persistent massive dysesthesia of the upper extremities, surgery was performed within the first week. A longitudinal approach at the medial border of the right sternocleidomastoid muscle was performed in order to intraoperatively assess adjacent segments of the cervical spine. A segment was defined clinically as “unstable in extension,” if there was a discontinuity of the ALL combined with a stripping of the disc from the adjacent end plate. The segment with the myelon edema was decompressed and fused in all cases, while adjacent segments were addressed in the presence of segmental instability.
All MRI scans of all patients were re-assessed by a radiologist with specialization in musculoskeletal MRI and high experience in the assessment of spinal pathologies for the purpose of this study after completion of treatment. He was given the same clinical information as the radiologist on call but was blinded to the intraoperative assessment of cervical instability.
The statistical analysis was performed using SPSS 24.0 (Chicago, IL, USA). Metric scaled data are reported as arithmetic mean ± standard deviation. Categorial data are reported as absolute frequencies and relative distribution in percent. MRI and intraoperative findings were compared, and sensitivity and specificity of MRI for detecting surgically verified hyperextension instability were calculated based on segments (n = 25) as well as patients (n = 22).
Twenty-three patients with a mean age of 62.7 ± 14.8 years (range 38–87 years) were included in this study. There were 20 men and 3 women. CT and MRI scans showed relevant degeneration and narrowing of the spinal canal in all patients. The center of the hyperintense spinal cord signal as assessed by MRI was located at C3/4 in 8 patients (34.8%), at C4/5 in 2 patients (8.7%), at C5/6 in 11 patients (47.8%), and at C6/7 in 2 patients (8.7%). There were no patients with more than one spinal cord signal.
Intraoperative findings revealed hyperextension instability in 22 patients (95.7%), while there were no signs of segmental instability in one patient (4.3%). Overall, 25 spinal segments with hyperextension instability due to anterior tension band disruption were observed intraoperatively in 22 patients. Segmental instability was found at C3/4 in 9 of 25 cases (36%), at C4/5 in 2 cases (8%), at C5/6 in 10 cases (40%), at C6/7 in 3 cases (12%), and at C7/Th1 in one case (4%).
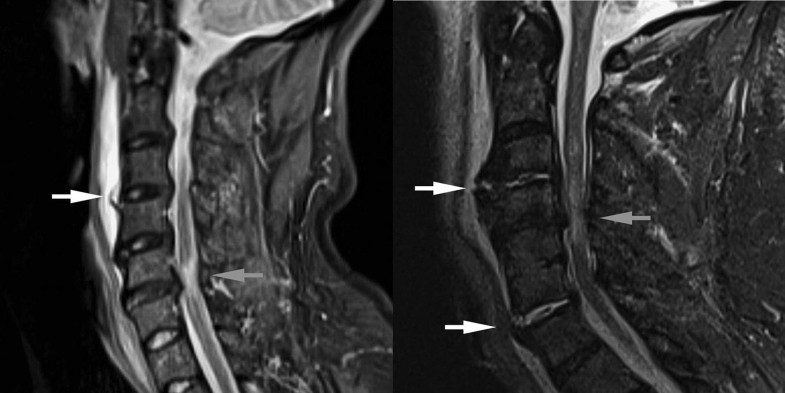
The comparison between intraoperative and MRI findings showed that the radiologist on call correctly assessed segmental hyperextension instability in 15 of 25 spinal segments with surgically verified hyperextension instability (sensitivity: 0.60), while the specialized MRI radiologist was correct in 22 of 25 segments (sensitivity: 0.88). Moreover, the radiologist on call correctly identified segmental hyperextension instability in 15 of 22 patients (sensitivity: 0.68) and the MRI radiologist in 19 of 22 patients (sensitivity: 0.86). Neither the radiologist on call nor the MRI radiologist assessed a segment as unstable in extension, which showed to be stable intraoperatively and during the further postoperative course (specificity: 1.00). Additionally, the PLL was described as intact in all cases by both radiologists, which matched the intraoperative findings.The topographic relations between the spinal cord signal and the intraoperative findings are shown in Table 1. The spinal cord signal and segmental instability were located at the same segment in 17 patients (73.9%, Fig. 1), whereas segmental instability and spinal cord signal were found at different levels in two patients (8.7%, Fig. 2a). In two patients (8.7%), segmental instability and spinal cord signal were located at the same segment, but an additional segmental instability was observed at an adjacent segment. In one patient (4.3%), the cord signal was located at C4/5, whereas segmental instabilities were found at C3/4 and C6/7 (Fig. 2b).Table 1
Topographic relations between spinal cord signals on MRI and intraoperative findings
| Number of patients | Segmental level of cord signal (CS) and segmental instability (SI) |
|---|---|
| 17 | CS and SI at the same spinal segment |
| 2 | CS and SI at the same segment + additional SI at an adjacent segment |
| 2 | CS and SI at different segments |
| 1 | CS at one segment and SI at two adjacent segments |
| 1 | CS without SI |
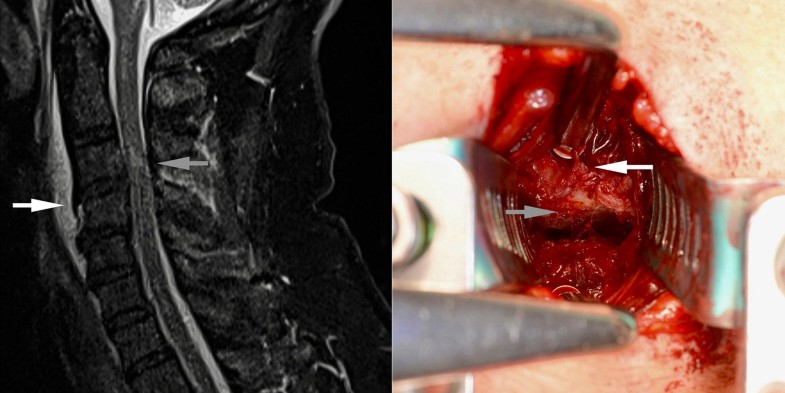
Our data show that:
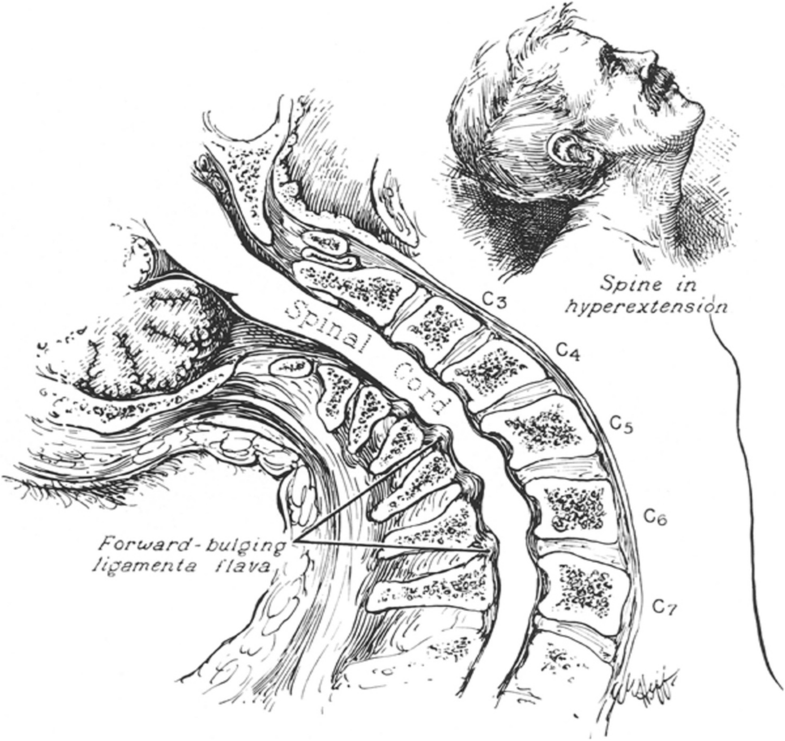
TCCS does not represent a recently defined clinical syndrome, but the first case report of TCCS rather dates back to 1887 [22]. In 1954, Schneider et al. [4] reported on a case series of 15 patients (since then described as “Schneider’s syndrome” by some authors). It was stated that TCCS occurs in three situations: (a) fractures and dislocations of the cervical spine, (b) sudden protrusion of an intervertebral disc with consecutive compression of the spinal cord, and (c) hyperextension of the cervical spine without segmental instability in patients with preexisting degenerative conditions of the cervical spine (spondylotic TCCS). In the latter group of patients, radiographs were described as showing no signs of fractures or instability, whereas CT and MRI scans were not available in 1954. Accordingly, Schneider et al. stated that spondylotic TCCS occurs without apparent injuries of the cervical spine by forward bulging of the ligamenta flava into the spinal canal resulting in spinal cord compression (Taylor mechanism [22], Fig. 3).
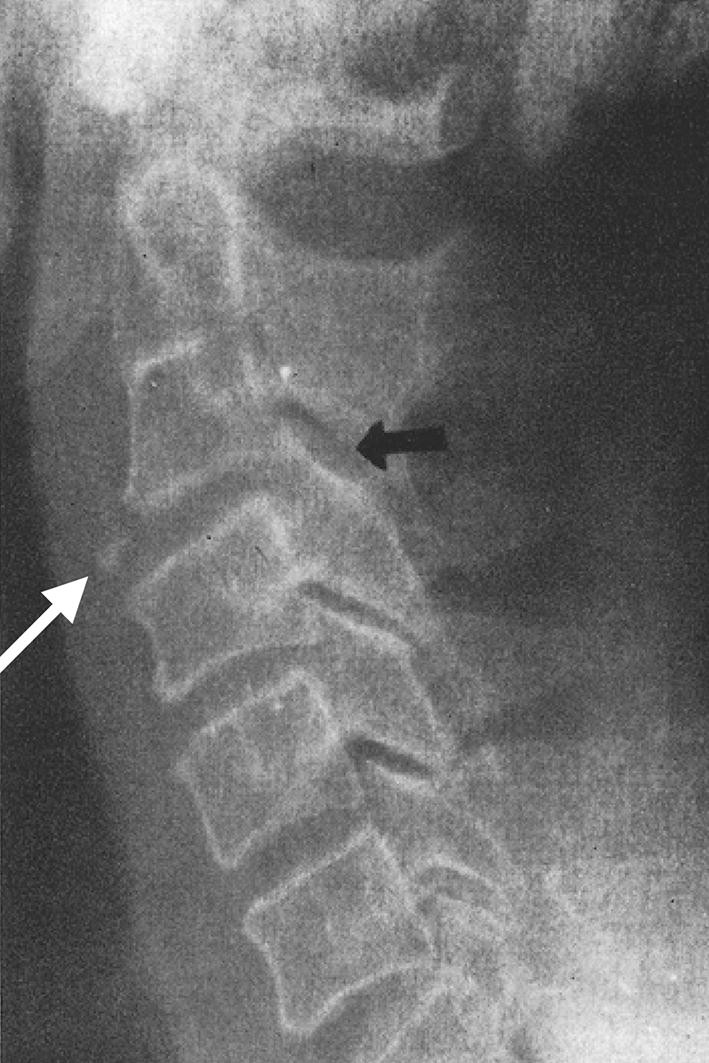
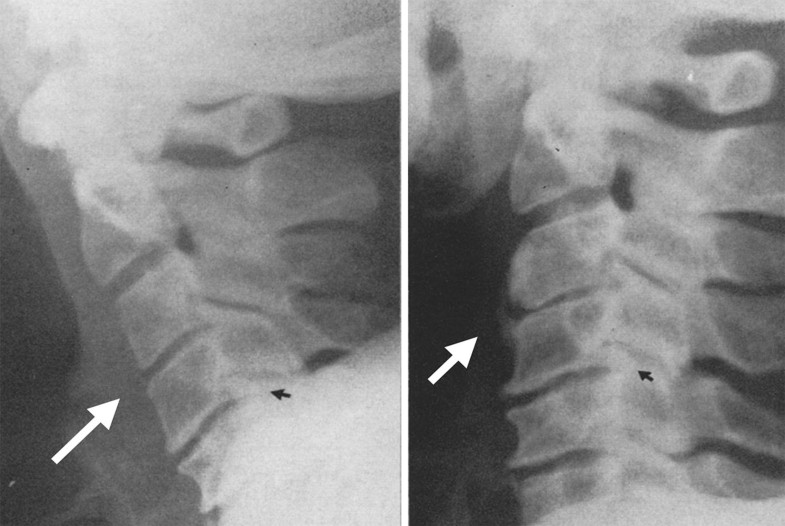
CT and MRI scans allow for the diagnosis of more subtle injuries of the cervical spine nowadays. CT scans represent the gold standard imaging technique for the assessment of fractures and subluxations, while MRI is the method of choice for the evaluation of ligamentous and extraneural soft tissue injuries as well as for the assessment of the spinal cord [21, 23]. Despite continuous advances in imaging during the last decades, spondylotic TCCS is still considered to result from compression of the spinal cord without acute discoligamentous instability of the cervical spine by several authors [3, 7, 8, 11, 13, 14]. According to the results of our study, however, spondylotic TCSS mainly results from hyperextension instability of the cervical spine following disruption of the anterior tension band. Interestingly, re-assessing radiographs presented in the hallmark paper by Schneider et al. [4] in 1954 supports our hypothesis by showing subtle signs of hyperextension instability in two cases (Figs. 4, 5).
The aim of the present study was not to prove the Taylor mechanism of spondylotic TCCS without instability to be generally wrong or irrelevant. The goal of this study rather was to increase the awareness for hyperextension instability in patients with TCCS without apparent instability on CT scans. We interpret our findings as follows: Our results are based on intraoperative findings as a reference for the assessment of instability and indications for surgery followed institutional guidelines as described in the methods section. Patients with less severe neurological impairment or complete remission within the first days were therefore treated nonoperatively and were excluded from this study. It is reasonable to assume that spinal cord compression was minor in these patients, since recent studies have revealed a correlation between the severity of neurological impairment and stability of the cervical spine [3, 24]. Accordingly, mild TCCS and TCCS with rapid improvement may occur in patients with stable cervical spines and may be attributed to the Taylor mechanism of spondylotic TCCS. In patients with profound TCCS, however, we consider hyperextension instability of the spondylotic spine as a relevant factor leading to increased compression of the spinal cord resulting in more severe neurological deficits. We therefore propose subclassification of spondylotic TCCS into “spondylotic TCCS without segmental instability” and “spondylotic TCCS with hyperextension instability.”
Accordingly, it is mandatory to actively confirm or rule out hyperextension instability in patients with spondylotic TCCS. MRI is the gold standard for the assessment of the spinal ligaments and the spinal cord [21, 23]. The ALL is the key structure for the assessment of hyperextension instability. An intact ALL appears as a hypointense band in all sequences and is hardly discernible from adjacent hypointense structures such as cortical bone or the annulus. A disruption of the ALL appears as focal discontinuity of the hypointense band frequently combined with elevation of the ligament from adjacent structures by fluid [23]. MRI findings were correlated with intraoperative findings as external reference following different types of cervical spine trauma in two studies [25, 26]. In 2006, Goradia et al. [25] used consensus reading of two radiologists of unknown experience and reported a sensitivity of MRI for ALL injuries of 0.71. A single senior trauma radiologist performed MRI reading in the study of Malham et al. [26] in 2009 with a sensitivity of 0.48 and a specificity of 1.00 for ALL injuries. The specificity of MRI was 1.00 in our study as well, while the sensitivity differed between the radiologist on call and the specialized MRI radiologist (0.61 and 0.88, respectively). Accordingly, hyperextension instability is detectable on MRI scans with an acceptable sensitivity in our opinion. It requires, however, high-resolution images with the entire bandwidth of available sequence and an experienced reader. Flexion–extension lateral radiographs may be an additional tool for the assessment of discoligamentous injuries of the cervical spine [24]. In patients with spondylotic TCCS, however, we do not recommend to perform lateral radiographs in maximum extension in order to avoid further spinal cord compression. Regarding the spinal cord, diffusion tensor imaging (DTI) in addition to conventional MRI may allow for a more detailed radiological assessment of spinal cord damage in the future [27]. DTI measures have been recently shown to correlate with the modified Japanese Orthopedic Association score in patients with cervical myelopathy [28] and may also provide useful information for treatment decision-making and predicting neurological outcome after TCCS.
Two further interesting findings of our study have to be discussed. First, the segmental level of spinal cord signal did not match the level of segmental instability in three patients. In these cases, the cord signal was found in the segment with the most pronounced spinal canal stenosis (Fig. 2). At first glance, this is a surprising finding which was not described in the literature yet. The spinal cord compression in spondylotic TCCS is the result of a pincer effect with the spinal cord being entrapped between the anterior spondylotic disc-osteophyte complex and the bulged posterior ligaments [29]. Spinal cord compression increases with increased extension of the cervical spine. The restricted ROM of a spondylotic cervical spine therefore may “protect” the spinal cord from major compression in patients without acute discoligamentous instability. Hyperextension instability, however, increases the ROM of the cervical spine in extension to a pathological level and thus results in more severe compression of the spinal cord occurring at the most stenotic level. Second, in three of twenty-three patients, hyperextension instability was observed at two segments of the lower cervical spine (Fig. 2). After high-energy trauma, bi- or multilevel injuries of the spine are not an uncommon finding and typically occur in different regions of the spinal column [30, 31]. For example, combined injuries of the upper and lower cervical spine as well as combinations of injuries of the cervical and thoracic spine have been frequently described. The patients in this study, however, sustained low-energy hyperextension trauma of the cervical spine, which resulted in ALL rupture and disc avulsion in the most brittle segment of the lower cervical spine in 22 patients. Further hyperextension in this segment might have been stopped by both the PLL and the intact sclerotic facets according to our interpretation. Continuing hyperextension forces therefore might have resulted in disruption of the anterior tension band at an additional brittle segment in these three patients.
Indications and timing for surgical decompression after spondylotic TCCS are controversially discussed in the literature. While Schneider et al. advised against any surgical intervention (posterior laminectomy and transdural discectomy at that time), there is increasing evidence from several recent studies that anterior decompression and fusion results in favorable neurological outcome [1, 2, 8, 10, 11, 13, 32]. Although early (< 24 h after injury) surgical decompression has been reported to be associated with improved neurological outcome after traumatic spinal cord injury (tSCI) [33, 34, 35], a recent web-based survey among Dutch surgeons found that patients with TCCS (and particularly those without spinal column and/or discoligamentous injury) are preferably treated less urgently than those with other forms of incomplete tSCI [36]. Our current institutional guidelines are based on an expert consensus [7], which recommends early surgical decompression in patients with AIS grade C and worse as well as initial observation of neurological recovery in less severe cases and optional surgery at a later date. In addition to these recommendations, we consider hyperextension instability as an indication for decompression and ACDF irrespective of the severity of neurological impairment. Nonoperative treatment may result in repetitive hyperextension at the injured segment and resulting compression of the spinal cord during the healing period even with the use of a cervical orthosis, which may impair neurological recovery.
Several limitations of this study need to be noted. First, this is a retrospective study with all limitations associated with this study design. Second, the number of patients included is relatively small due to the strict inclusion criteria. The latter, however, allowed for a homogeneous collective of patients with profound spondylotic TCCS and well-documented intraoperative findings, although TCCS in general represents a heterogeneous syndrome. Nevertheless, patients with mild TCSS or rapid neurological improvement within the first hours were treated nonoperatively and were therefore excluded from this study due to the unavailability of intraoperative findings. Third, the assessment of postoperative neurological recovery was not part of this study.
The findings of this study indicate that hyperextension instability due to anterior tension band failure is highly common in patients presenting with profound spondylotic TCCS after hyperextension trauma. In view of these findings, presence or absence of segmental instability should be thoroughly assessed by high-quality MRI and an experienced radiologist prior to therapeutic decision-making. In spondylotic TCSS with instability, early surgical decompression and stabilization of the spinal segment(s) with hyperextension instability and/or spinal cord signal is considered most appropriate.
Open access funding provided by University of Innsbruck and Medical University of Innsbruck.
The authors declared that they have no conflict of interest.
586_2018_5796_MOESM1_ESM.pptx (138 kb)Supplementary material 1 (PPTX 138 kb)
© The Author(s) 2018
Open AccessThis article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.

C/ San Pedro de Mezonzo nº 39-41
15701 – Santiago de Compostela
Teléfono: +34 986 417 374
Email: secretaria@sogacot.org
Coordinador del Portal y Responsable de Contenidos: Dr. Alejandro González- Carreró Sixto